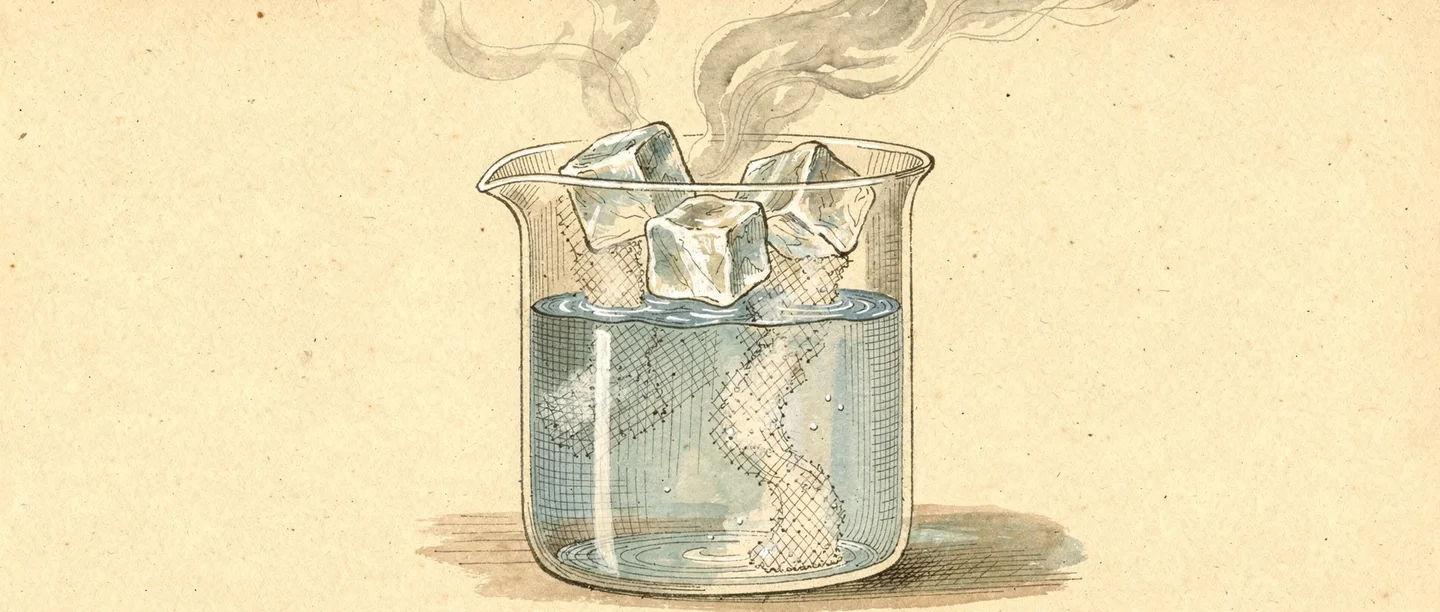What are the states of matter — the three main states
The states of matter are the different forms in which matter can exist. They are determined by how particles are arranged and how much energy they have. There are three states of matter that students encounter in everyday life:
Solid
In a solid, particles are packed tightly together in a fixed arrangement. They vibrate in place but cannot move freely. This gives solids a definite shape and a definite volume. Examples: ice, rock, wood, metal.
Liquid
In a liquid, particles are close together but not rigidly fixed. They can slide past each other. This means liquids have a definite volume but no fixed shape — they take the shape of whatever container holds them. Examples: water, juice, mercury.
Gas
In a gas, particles are widely spread out and move freely and rapidly in all directions. Gases have no fixed shape and no fixed volume — they expand to fill any container. Examples: air, steam, oxygen, carbon dioxide.
A fourth state, plasma, exists in stars and lightning — rarely encountered in everyday life.
In essence, it is a question about how particles behave under different conditions of temperature and pressure.

How matter changes state — from solid to gas and back
Matter does not stay in one state forever. Adding or removing heat energy causes the states of matter to change. These transitions have specific names:
- Melting — solid becomes liquid (e.g., ice melting into water at 0°C)
- Freezing — liquid becomes solid (e.g., water freezing at 0°C)
- Evaporation / Boiling — liquid becomes gas (e.g., water boiling to steam at 100°C)
- Condensation — gas becomes liquid (e.g., steam condensing on a cold mirror)
- Sublimation — solid becomes gas directly without passing through liquid (e.g., dry ice — solid CO₂ — sublimes at room temperature)
Changes of state are physical changes, not chemical ones. The substance remains the same — only the arrangement of its particles changes. When steam condenses into water, the water molecules are identical to those that evaporated.
Each substance has a fixed melting point and boiling point — constants used to identify materials.
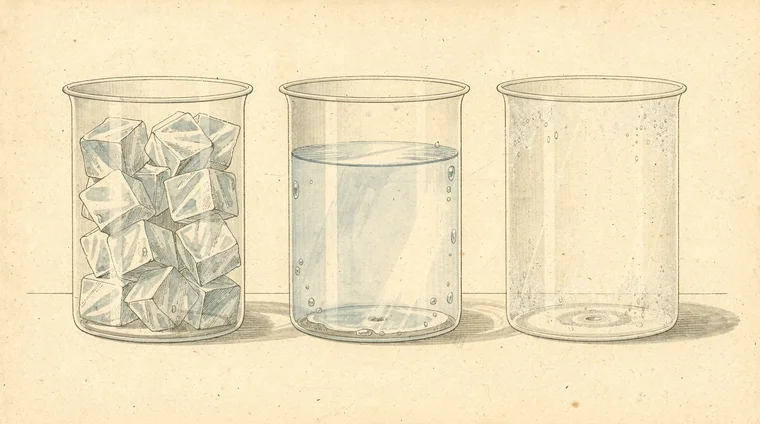
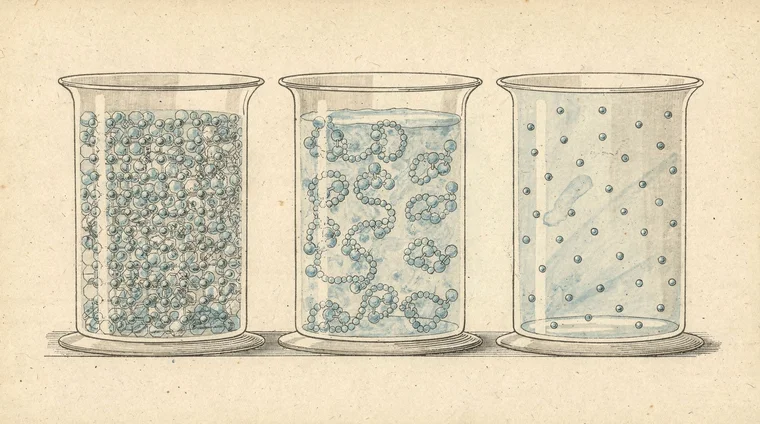
Particle theory — why the states of matter behave differently
The particle model of matter explains the states of matter at a microscopic level. The key ideas are:
- All matter is made up of tiny particles (atoms or molecules).
- Particles are always moving.
- Particles attract each other; the strength of attraction varies.
- The higher the temperature, the more energy the particles have — and the faster they move.
In a solid, particles vibrate around fixed positions. The attractive forces are strong enough to keep them locked in place.
In a liquid, particles have more energy. They move enough to slide past each other, but attractive forces still keep them close.
In a gas, particles have so much energy that they overcome attractive forces entirely. They move rapidly and independently, spreading out to fill their container.
When you heat a solid, you give its particles more energy. Eventually they have enough energy to break free from their fixed positions — melting occurs. Add more heat and liquid particles gain enough energy to escape from the liquid surface entirely — evaporation or boiling occurs.
This particle model is the foundation of chemistry and physics at secondary school. Everyday examples — a drying puddle, steam over a hot drink — help make these concepts tangible. See For parents for more activity ideas.
Frequently asked questions
- Is water the only substance that can exist in all three states of matter in everyday conditions?
- Water is the most familiar example — ice, liquid water, and steam are all the same substance at different temperatures. But most substances can exist in all three states; they just require different temperatures. Iron, for instance, is liquid at 1538°C and a gas above 2862°C.
- Why does a gas expand to fill its container?
- Gas particles move randomly and rapidly in all directions with no significant attractive forces between them. They spread out evenly to fill any space available. Open a bottle of perfume and gas molecules spread throughout the room — which is why you can smell it from across the room.
- What happens to mass when a substance changes state?
- Mass is conserved during changes of state. When ice melts into water or water evaporates, no matter is gained or lost — the particles are just rearranging. This is a key principle: in a closed system, total mass never changes regardless of changes of state.
- At what age do children learn about the states of matter in school?
- Solid, liquid, and gas are typically introduced around ages 7–9 (grades 2–4) using examples like ice, water, and steam. Particle theory is introduced around ages 11–13 (grades 6–8), explaining why changes of state happen.