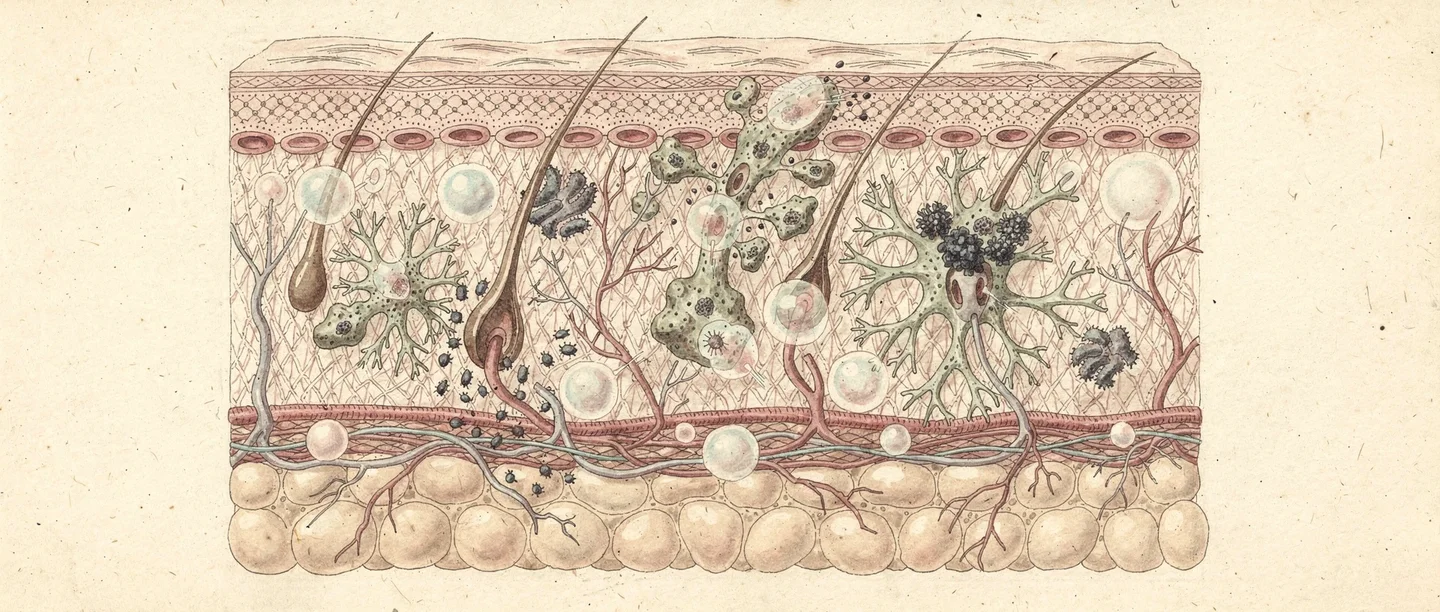
The innate immune system: your instant defence
The moment a pathogen enters your body — through a cut, or inhaled air — the immune system's first line of defence activates within minutes. This rapid, non-specific response is called the innate immune system.
Tissue cells called macrophages patrol constantly. These cells carry toll-like receptors — surface proteins that recognise molecular patterns unique to bacteria. Examples include lipopolysaccharides on bacterial membranes, and flagellin in bacterial tails. When a macrophage detects these patterns, it sounds the alarm by releasing chemical signals called cytokines.
Cytokines trigger inflammation: blood vessels widen, immune cells flood the area, and your temperature may rise. Each of these responses makes the environment harder for pathogens to survive.
Other innate defences include natural killer cells, which destroy virus-infected host cells. Complement proteins circulate in blood and punch holes through bacterial membranes.
The innate immune system is fast but not precise. It buys time for the body's second, more targeted system to activate. Parents can explore the for parents guide for recommended books and resources.
The foundational reference for immunology is Janeway's Immunobiology, freely available via NCBI.
The adaptive immune system: learning to fight back
The innate immune system hands off to a slower but far more powerful system — the adaptive immune system. This is what makes vaccines possible and what gives you long-lasting protection after recovering from an infection.
Before birth, your body generates around one billion distinct receptor shapes on T-cells and B-cells — entirely at random. When a pathogen enters the body, at least one receptor shape is highly likely to match part of its surface — the antigen.
When a T-cell or B-cell finds a matching antigen, it divides rapidly. This produces millions of identical copies — clonal expansion. Some of these copies become effector cells that attack the infection right now. Others become memory cells that persist in the body for decades, sometimes for life.
When the same pathogen enters the body a second time, those memory cells recognise it immediately. The response is orders of magnitude faster and stronger — often clearing the infection before any symptoms appear. This is how vaccines work. They introduce a harmless version of a pathogen so the immune system builds memory cells — without the risk of real infection.
This process connects to what DNA is — receptor diversity is generated by deliberate DNA shuffling unique to immune cells.
Did you know?
-
Macrophages recognise bacterial patterns such as lipopolysaccharides and flagellin via toll-like receptors, triggering inflammation within minutes of infection.
Janeway's Immunobiology -
Around one billion distinct T-cell and B-cell receptor shapes are generated randomly before birth, making it highly probable the immune system can recognise any pathogen it encounters.
Janeway's Immunobiology -
B-cells undergo affinity maturation in lymph nodes — a process of rapid evolution that makes antibodies progressively more precise as an infection continues.
Victora & Nussenzweig 2012, Nature Reviews Immunology -
Measles requires around 95% of a population to be immune to prevent transmission. When vaccination rates fall below this threshold, outbreaks become likely.
Anderson & May 1985, Nature -
The hygiene hypothesis suggests that reduced early-life exposure to parasites and microbes may leave the immune system prone to attacking harmless substances — contributing to the rise of allergies.
Strachan 1989, BMJ
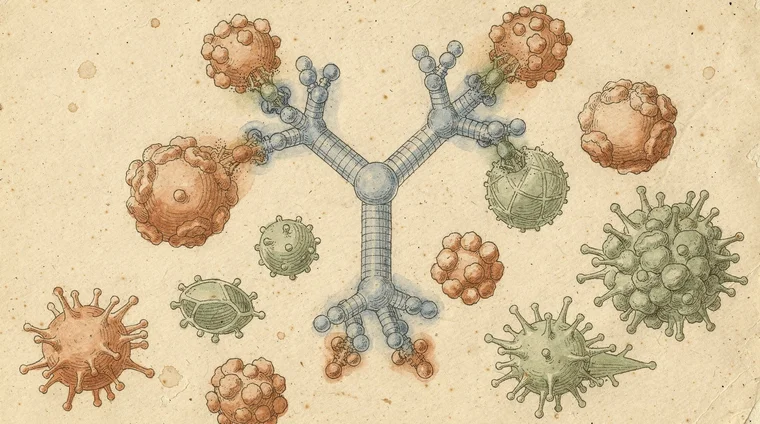
Antibodies and herd immunity
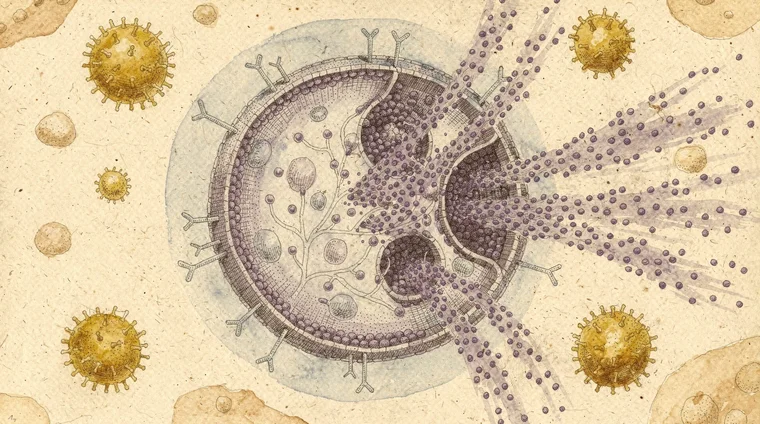
B-cells are the immune system's factories for a special class of proteins called antibodies. Each antibody is Y-shaped. Its two arms bind a specific antigen — neutralising it or marking it for destruction.
When B-cells first encounter a matching antigen, they migrate to lymph nodes. There they undergo affinity maturation — rapid mutation and selection that makes antibodies progressively more precise. This is essentially evolution happening inside your lymph nodes over the course of an infection.
Herd immunity and vaccines
Because vaccination primes the immune system with memory cells, entire communities can be protected. This extends to those who cannot be vaccinated — newborns, immunocompromised patients, and people with severe allergies to vaccine components.
This protection is called herd immunity. It works because if enough people are immune, a pathogen cannot find enough susceptible hosts to spread. The required proportion varies by pathogen. For measles — one of the most contagious diseases known — approximately 95% of the population must be immune to stop transmission. For polio, the threshold is around 80%.
When vaccination rates fall below these thresholds, pathogens find gaps in the population's defences and outbreaks follow. High and consistent vaccination coverage matters. It protects not just individuals, but those who rely on others' immunity.
For a broader view, what is evolution explains why pathogens evolve to evade immune defences — and why adaptive immunity matters.
For the original research, see Victora & Nussenzweig (2012) on germinal centres and antibody maturation.
When the immune system goes wrong: allergies
The immune system is not infallible. In some people, it mounts a response against harmless substances — pollen, peanut proteins, pet dander, or latex. This is an allergy.
The mechanism involves a class of antibodies called IgE. In people prone to allergies, IgE antibodies form against harmless antigens. They attach to mast cells distributed throughout the body. When the allergen appears again, it cross-links those IgE antibodies. This triggers mast cells to release histamine and other chemicals.
Histamine causes the familiar symptoms of an allergic reaction: blood vessels dilate, tissues swell, airways may narrow. In a localised reaction — sneezing from pollen, itching from a bee sting — this is uncomfortable but not dangerous. In severe cases, anaphylaxis can occur — a systemic reaction with a sudden blood pressure drop and airway constriction. Without immediate treatment, it can be life-threatening.
Why allergies have increased
One influential explanation is the hygiene hypothesis, proposed by David Strachan in 1989. Strachan observed that children in larger families — with more early exposure to infections — were less likely to develop allergies. The hypothesis holds that reduced early-life exposure to parasites and microbes may leave the immune system under-challenged. It becomes more prone to treating harmless substances as threats.
This does not mean hygiene is harmful. Rather, it points to the value of early exposure to diverse natural environments. Research into the gut microbiome continues to explore these connections.
For cellular biology context, what is a cell explains how immune cells like mast cells and B-cells develop and function.
Frequently asked questions
- How long does it take for the immune system to respond to an infection?
- The innate immune system responds within minutes to hours — macrophages can detect bacterial patterns almost immediately. The adaptive immune system takes longer: it typically takes 4–7 days for T-cells and B-cells to multiply and mount a full targeted response. This is why symptoms of many illnesses peak around day 3–5 and then improve as the adaptive response gains strength.
- Can the immune system become weaker over time?
- Yes. The immune system naturally declines with age — a process called immunosenescence. Older adults produce fewer naive T-cells and B-cells, so responses to new pathogens can be slower and weaker. Chronic stress, poor sleep, malnutrition, and some diseases can also suppress immune function. This is one reason vaccines are particularly important for older people, and why higher-dose formulations are available for some vaccines targeted at elderly populations.
- Why do some people get allergies and others do not?
- Genetics plays a significant role — if one parent has allergies, a child has roughly a 30–40% chance of developing them; if both parents do, the risk rises to 60–70%. But environment matters too. Early childhood exposure to diverse microbes, farm animals, and natural environments is associated with lower allergy rates. Diet, gut microbiome composition, and the timing of first exposure to certain foods also influence whether the immune system learns to tolerate them.
- How do vaccines teach the immune system without causing disease?
- Vaccines introduce the immune system to an antigen — typically a weakened or inactivated pathogen, a fragment of its surface, or genetic instructions for producing a harmless piece of it — without the pathogen being able to replicate and cause illness. The immune system generates memory cells just as it would after a real infection. If the actual pathogen is encountered later, those memory cells enable a fast, strong response that clears the infection quickly.