The Structure of an Atom
An atom has two main regions. At the centre is the nucleus, which contains protons (positively charged) and neutrons (no charge). Surrounding the nucleus is a cloud of electrons (negatively charged) arranged in shells.
The number of protons in an atom's nucleus is the atomic number — the unique identifier for that element. Hydrogen atoms have 1 proton. Carbon has 6. Gold has 79. Change the number of protons and you have a different element entirely.
Electrons and chemical bonding
The outermost shell of electrons determines how atoms bond with other atoms. Atoms with partly filled outer shells readily share or transfer electrons to reach a stable arrangement. Noble gases, whose outer shells are already full, are almost completely unreactive.
Ions
Atoms can gain or lose electrons to become ions — electrically charged atoms. Sodium loses one electron to form Na⁺. Chlorine gains one to form Cl⁻. These opposite charges attract each other, forming ionic bonds — the bonds that hold salt crystals together. The periodic table organises all elements by their proton count.

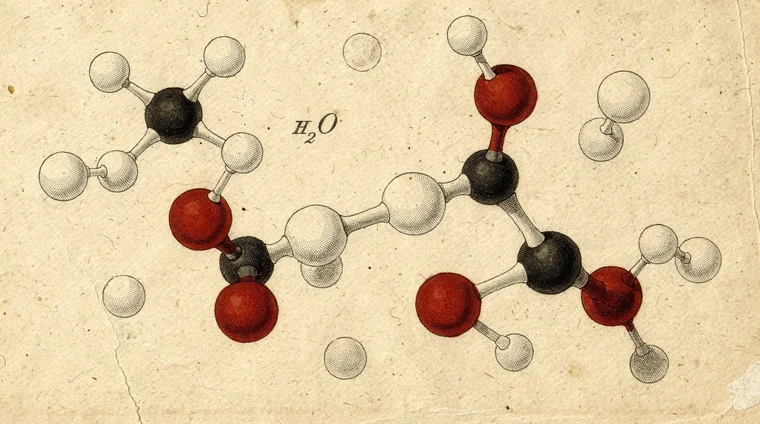
How Atoms Form Molecules and Compounds
Elements rarely exist as single isolated particles. They bond together to form molecules — groups of two or more atoms held together by chemical bonds.
When atoms share electrons, they form covalent bonds. Water (H₂O) is two hydrogen atoms bonded to one oxygen atom. Carbon dioxide (CO₂) is one carbon atom double-bonded to two oxygen atoms. The specific arrangement in a molecule determines its properties.
Elements and compounds
A substance made of only one type of atom is an element — pure iron, pure oxygen, pure gold. A substance where different elements are chemically bonded together is a compound. Table salt (NaCl) is a compound of sodium atoms and chlorine atoms. Compounds have entirely different properties from their component elements.
Mixtures
Atoms and molecules can also combine without forming new compounds — these are mixtures. Air is a mixture of nitrogen, oxygen, argon, and other molecules. Unlike compounds, mixtures can be separated by physical means such as filtration or evaporation.
Atoms: From Ancient Idea to Modern Science
The word atom comes from the Greek atomos, meaning 'uncuttable'. The philosopher Democritus proposed around 400 BCE that all matter was made of indivisible particles. But scientific atomic theory only emerged in the 19th century.
John Dalton formalised the theory in 1803: particles of each element were identical, and chemical reactions rearranged them without creating or destroying anything. J.J. Thomson discovered the electron in 1897. Ernest Rutherford showed in 1911 that matter has a tiny, dense nucleus surrounded by mostly empty space.
Quantum mechanics and atoms
The modern model of atoms comes from quantum mechanics. Electrons do not orbit the nucleus in fixed paths — they occupy probability clouds called orbitals. This model explains why elements emit light only at specific wavelengths and why the periodic table has the structure it does.
Atoms in everyday life
Every chemical reaction involves particles rearranging. Burning fuel, digesting food, and charging a battery are all processes in which particles form new bonds or break old ones. Understanding DNA depends on how carbon, nitrogen, oxygen, and phosphorus bond into precise molecular structures.
Frequently asked questions
- How small is an atom?
- Atoms are about 0.1 to 0.5 nanometres across — a nanometre is one billionth of a metre. A million atoms lined up would span one millimetre. The nucleus at the centre is 100,000 times smaller than the whole atom. If an atom were the size of a football stadium, its nucleus would be roughly the size of a marble.
- Can atoms be created or destroyed?
- In ordinary chemistry, they are neither created nor destroyed — only rearranged. This is the law of conservation of mass. In nuclear reactions, they can be split (fission) or fused (fusion), releasing enormous energy. These processes convert a tiny amount of mass into energy, as described by Einstein's equation E = mc².
- Are all atoms of the same element identical?
- Not quite. All atoms of the same element have the same number of protons, but neutron counts can vary. These variants are called isotopes. Carbon-12 has 6 protons and 6 neutrons; carbon-14 has 6 protons and 8 neutrons. Carbon-14 is radioactive and is used in radiocarbon dating to estimate the age of ancient materials.
- What holds an atom's nucleus together?
- The nucleus is held together by the strong nuclear force, which overcomes the electrical repulsion between positively charged protons. Electrons are bound near the nucleus by electromagnetic attraction. If the nucleus becomes too large or has an unfavourable ratio of protons to neutrons, it becomes unstable and radioactively decays.
