The Nature of Light: Wave and Particle
Light behaves in two seemingly contradictory ways: as a wave and as a particle. This is called wave-particle duality and is one of the central ideas of quantum physics.
As a wave, light has a wavelength (distance between peaks) and a frequency (cycles per second). Visible radiation — what our eyes can detect — has wavelengths between about 380 nanometres (violet) and 700 nanometres (red).
As a particle, light travels in discrete packets of energy called photons. Each photon carries a fixed amount of energy determined by its frequency. High-frequency radiation (ultraviolet, X-rays) carries more energy per photon than low-frequency radiation (infrared, radio waves).
The electromagnetic spectrum
Visible light is just a small portion of the electromagnetic spectrum. Radio waves, microwaves, infrared, ultraviolet, X-rays, and gamma rays are all electromagnetic radiation. All travel at the same speed, differing only in wavelength and frequency. The study of how sound travels contrasts with light: sound needs a medium, but it does not.

How Light Behaves: Reflection, Refraction, and Colour
Light interacts with matter in predictable ways that explain much of what we see.
Reflection
When a ray strikes a smooth surface, it bounces back at the same angle it arrived — this is reflection. Mirrors reflect almost all radiation that hits them. Rough surfaces scatter light in many directions — which is why paper looks white but not mirror-like.
Refraction
When radiation passes from one medium into another — from air into glass or water — it changes speed and bends. This is refraction. A pencil in a glass of water appears bent because rays from it refract as it moves from water to air. Lenses use refraction to focus radiation.
Colour
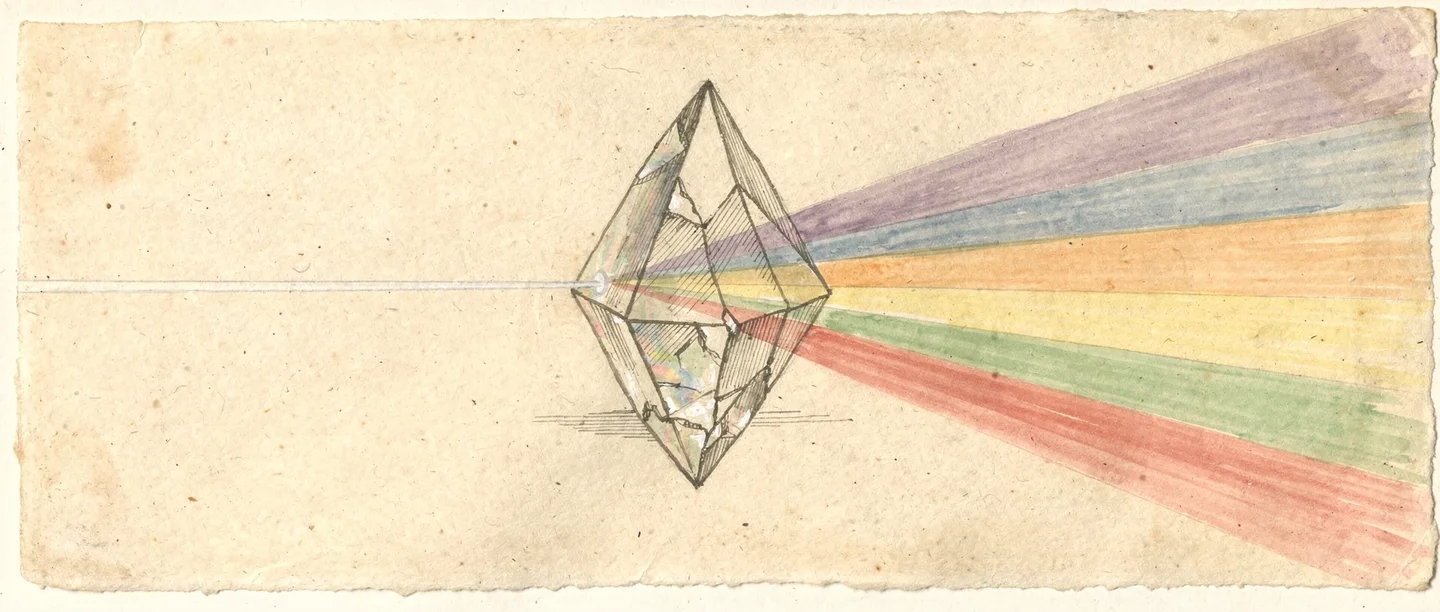
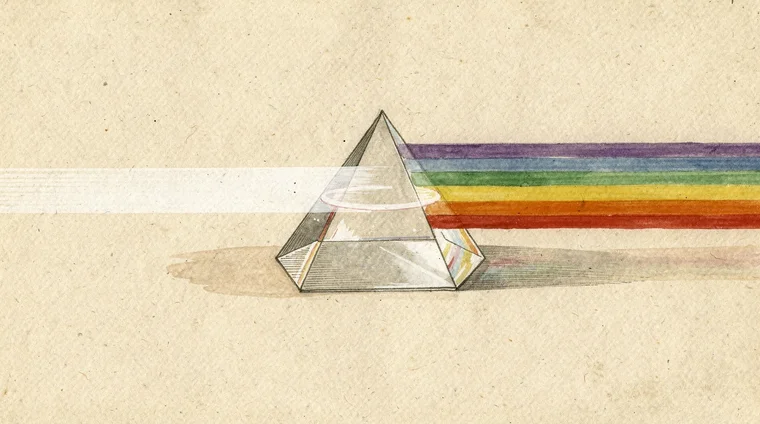
Sunlight is a mixture of all visible wavelengths. A prism separates these wavelengths by refracting each colour by a different amount, producing a rainbow spectrum. Objects appear coloured because they absorb some wavelengths and reflect others. A red apple absorbs most light but reflects red wavelengths back to your eyes.
The Speed of Light and Its Importance
Electromagnetic radiation travels at approximately 299,792,458 metres per second in a vacuum — a constant so fundamental it is given its own symbol: c. Nothing with mass can reach this speed, and the speed of light sets an absolute limit on how fast information can travel.
Why the speed of light matters
Einstein's special relativity, published in 1905, showed that this speed is the same for all observers regardless of how fast they or the source of light is moving. The consequences were extraordinary: moving clocks tick more slowly, objects contract in length, and mass and energy are equivalent (E = mc²).
Light in technology
Fibre-optic cables carry internet data as pulses of radiation. Information travels at close to the speed of light over long distances. Lasers concentrate light into precise beams used in surgery, manufacturing, and reading barcodes. Solar panels convert radiant energy directly into electricity using the photoelectric effect — the same quantum phenomenon that helped earn Einstein his Nobel Prize.
Frequently asked questions
- Why does light travel so fast?
- The speed of light in a vacuum is a fundamental constant of nature — it does not have a cause in the usual sense. It emerges from the properties of electric and magnetic fields. In any denser medium, light slows: it travels at about 75% of its vacuum speed in glass, and about 45% in diamond.
- Can light be slowed down or stopped?
- Light slows whenever it passes through a material. In glass it travels at about 200,000 km/s rather than 300,000 km/s. In 1999, scientists cooled a gas of atoms to near absolute zero and slowed light to just 17 metres per second. Stopping it completely remains impossible under normal conditions, but very slow light has been achieved in laboratory settings.
- What is ultraviolet light and why is it harmful?
- Ultraviolet (UV) radiation has wavelengths shorter than visible radiation, carrying more energy per photon. UV radiation can damage DNA in skin cells, causing sunburn and increasing the risk of skin cancer. The ozone layer absorbs most UV-B and UV-C from the sun. Sunscreen absorbs or reflects UV before it reaches the skin.
- How do our eyes detect light?
- The eye contains photoreceptors in the retina. Cone cells detect colour in bright conditions — three types respond to red, green, and blue wavelengths. Rod cells detect intensity in dim light but cannot distinguish colour. Signals travel via the optic nerve to the brain, which constructs the image we perceive.